Publications Laboratory of Cellular Optics II
G.J. Dickey, K. Bian, S.U. Islam, H.R. Khan, S. Rohr, H. Mao.
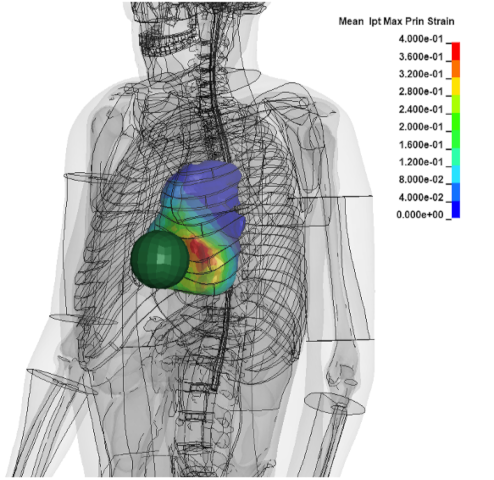
Advancing Commotio cordis Safety Standards Using the Total Human Models for Safety (THUMS). Ann Biomed Eng 2023 DOI
Advancing Commotio cordis Safety Standards Using the Total Human Models for Safety (THUMS). Ann Biomed Eng 2023 DOI
M. Rieger, C. Dellenbach, J. vom Berg, J. Beil-Wagner, A. Maguy &
S. Rohr.
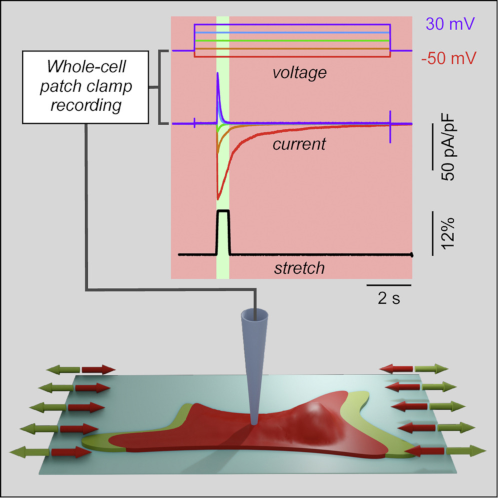
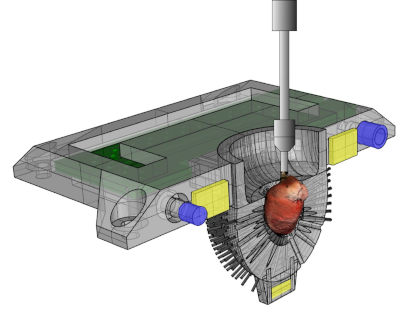
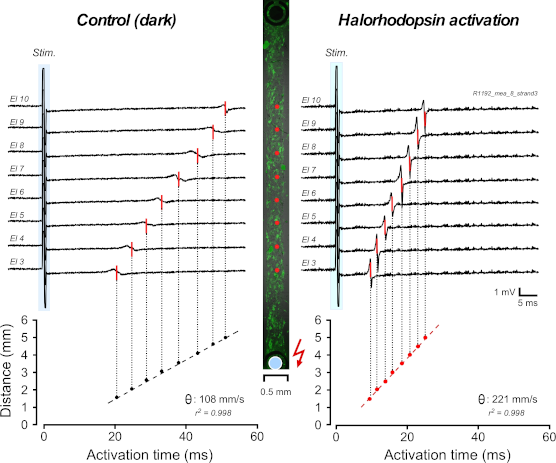
Enabling comprehensive optogenetic studies of mouse hearts by simultaneous opto-electrical panoramic mapping and stimulation. Nature Communications 2021 DOI
Enabling comprehensive optogenetic studies of mouse hearts by simultaneous opto-electrical panoramic mapping and stimulation. Nature Communications 2021 DOI
B.X.E. Desbiolles*, E. de Coulon*, N. Maïno, A. Bertsch, S. Rohr& and P. Renaud&, *: shared first authorship; &: shared last authorship.
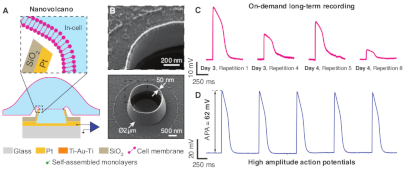
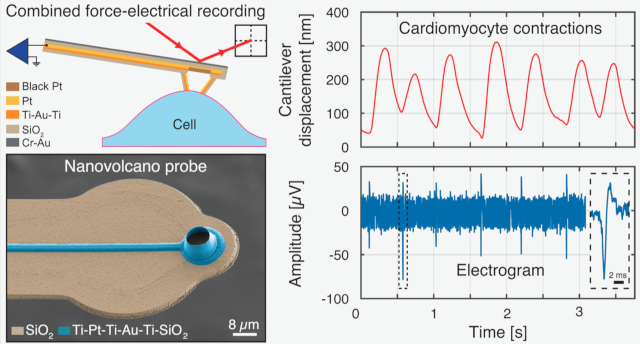
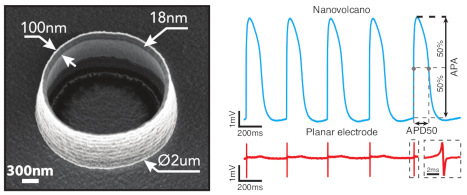
Nanovolcano microelectrode arrays: toward long-term on-demand registration of transmembrane action potentials by controlled electroporation. Microsyst Nanoeng 6, 67, 2020 DOI
Nanovolcano microelectrode arrays: toward long-term on-demand registration of transmembrane action potentials by controlled electroporation. Microsyst Nanoeng 6, 67, 2020 DOI
S.A. De Simone, S.Moyle, A. Buccarello, C. Dellenbach, J.P. Kucera and S. Rohr.
The Role of Membrane Capacitance in Cardiac Impulse Conduction: An Optogenetic Study With Non-excitable Cells Coupled to Cardiomyocytes. Front. Physiol 2020 DOI
The Role of Membrane Capacitance in Cardiac Impulse Conduction: An Optogenetic Study With Non-excitable Cells Coupled to Cardiomyocytes. Front. Physiol 2020 DOI
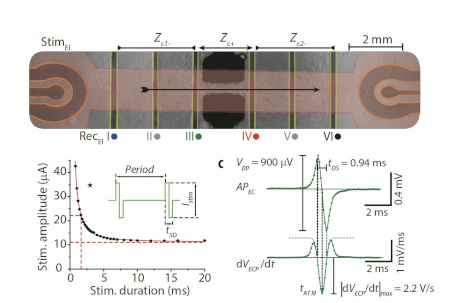
M. Imboden, E. de Coulon, A. Poulin, C. Dellenbach, S. Rosset,
H. Shea & S. Rohr.
High-speed mechano-active multielectrode array for investigating rapid stretch effects on cardiac tissue. Nature Communications 10:834, 2019 DOI
High-speed mechano-active multielectrode array for investigating rapid stretch effects on cardiac tissue. Nature Communications 10:834, 2019 DOI
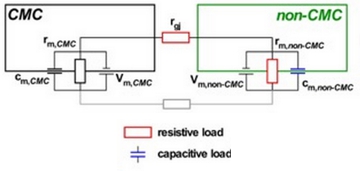
J.P. Kucera, S. Rohr, A.G. Kléber.
Review: Microstructure, cell-to-cell coupling, and ion currents as determinants of electrical propagation and arrhythmogenesis . Circ. Arrhythm. Electrophysiol. 10:e004665, 2017 DOI
Review: Microstructure, cell-to-cell coupling, and ion currents as determinants of electrical propagation and arrhythmogenesis . Circ. Arrhythm. Electrophysiol. 10:e004665, 2017 DOI
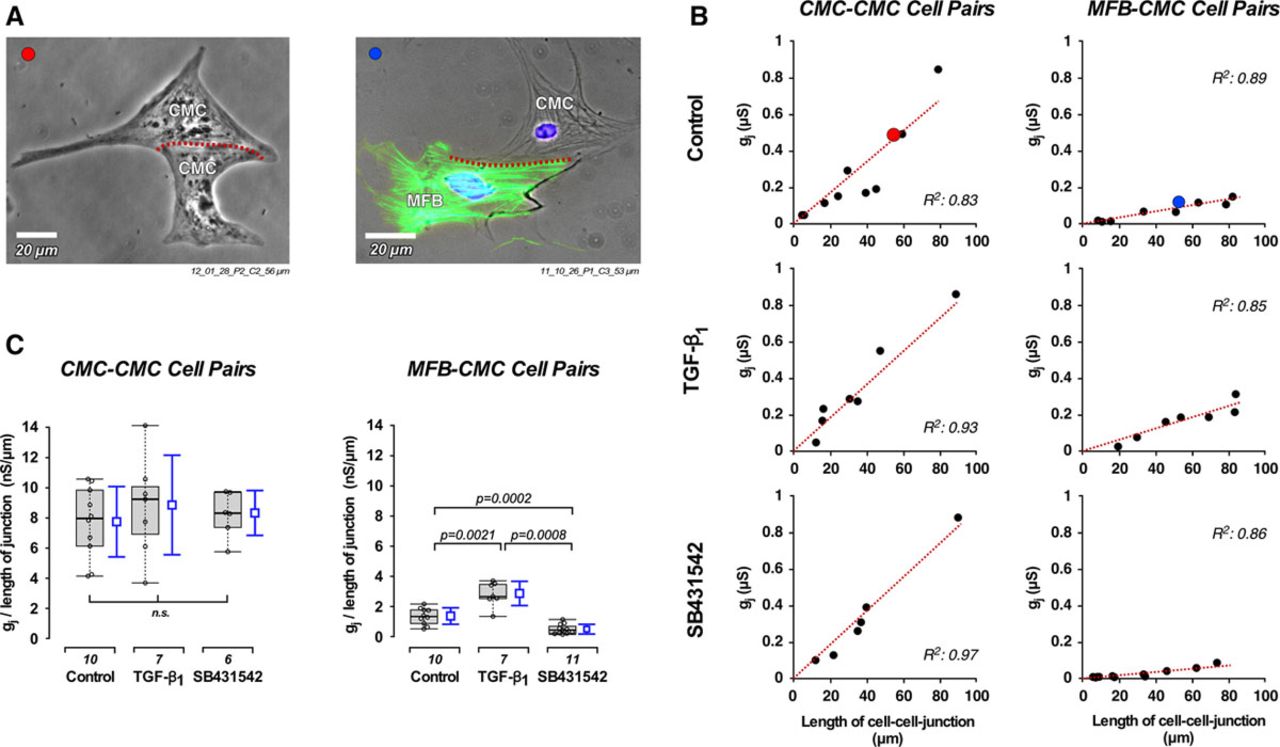
N Salvarani, A. Maguy, S.A. De Simone, M. Miragoli, F. Jousset, S. Rohr.
TGF-β1 (Transforming Growth Factor-β1) Plays a Pivotal Role in Cardiac Myofibroblast Arrhythmogenicity. Circ. Arrhythm. Electrophysiol. 10(5):e004567, 2017 DOI
TGF-β1 (Transforming Growth Factor-β1) Plays a Pivotal Role in Cardiac Myofibroblast Arrhythmogenicity. Circ. Arrhythm. Electrophysiol. 10(5):e004567, 2017 DOI
Perspectives from
Kuljeet Kaur, PhD; José Jalife, MD. Is TGF-β1 (Transforming Growth Factor-β1) an Enabler of Myofibroblast–Cardiomyocyte Cross Talk?. Circ. Arrhythm. Electrophysiol. 10:e005289, 2017 DOI
Perspectives from
G. Gabbiani. . F1000Prime 2017 DOI
F. Jousset, A. Maguy, S. Rohr, J.P. Kucera.
Myofibroblasts Electrotonically Coupled to Cardiomyocytes Alter Conduction: Insights at the Cellular Level from a Detailed In silico Tissue Structure Model. Front. Physiol 7:496, 2016 DOI
Myofibroblasts Electrotonically Coupled to Cardiomyocytes Alter Conduction: Insights at the Cellular Level from a Detailed In silico Tissue Structure Model. Front. Physiol 7:496, 2016 DOI
D. Brönnimann, T. Djukic, R. Triet, C. Dellenbach,
I. Saveljic, M. Rieger, S. Rohr, N. Filipovic, V. Djonov.
Pharmacological Modulation of Hemodynamics in Adult Zebrafish In Vivo. PLoS ONE 11(3): e0150948, 2016 DOI pdf
Pharmacological Modulation of Hemodynamics in Adult Zebrafish In Vivo. PLoS ONE 11(3): e0150948, 2016 DOI pdf
Perspectives from
M.H. Gollob. Toward Translation of Genomic Discovery to Clinical Efficacy in Atrial Fibrillation. JACC 68(17), 2016 DOI
F. Jousset, S. Rohr.
Optical recording of calcium currents during impulse conduction in cardiac tissue. Neurophotonics 2 (2), 2015 DOI
Optical recording of calcium currents during impulse conduction in cardiac tissue. Neurophotonics 2 (2), 2015 DOI
U. Schotten, S. Hatem, U. Ravens, P. Jaïs, F.U. Müller, A, Goette, S. Rohr, G. Antoons, B. Pieske, D. Scherr, A. Oto, B. Casadei, S. Verheule, D. Cartlidge, K. Steinmeyer, T. Götsche, D. Dobrev, J. Kockskämper, U. Lendeckel, L. Fabritz, P. Kirchhof, and A.J. Camm.
The European Network for Translational Research in Atrial Fibrillation (EUTRAF): objectives and initial results. Europace 2015 DOI
The European Network for Translational Research in Atrial Fibrillation (EUTRAF): objectives and initial results. Europace 2015 DOI
T. Grand, N. Salvarani, J. Jousset, S. Rohr.
Aggravation of cardiac myofibroblast arrhythmogeneicity by mechanical stress. Cardiovasc. Res. 104:489-500, 2014 DOI
Aggravation of cardiac myofibroblast arrhythmogeneicity by mechanical stress. Cardiovasc. Res. 104:489-500, 2014 DOI
S. Rohr.
Arrhythmogenic Implications of Fibroblast-Myocyte Interactions. Circ. Arrhythm. Electrophysiol. 5:442-452, 2012 DOI
Arrhythmogenic Implications of Fibroblast-Myocyte Interactions. Circ. Arrhythm. Electrophysiol. 5:442-452, 2012 DOI
S. Hatem, U. Schotten, S. Rohr, D. Dobrev, U. Ravens, A. Goette, B. Pieske, J. Kockskämper, U. Lendeckel, B. Casadei, P. Kirchhof, F.U. Müller, P. Jais, A. Oto, J.A. Camm.
The European Network for Translational Research in Atrial Fibrillation. Clin Invest 2(11): 1061-1067, 2012 DOI
The European Network for Translational Research in Atrial Fibrillation. Clin Invest 2(11): 1061-1067, 2012 DOI
S. Morel, Frias M.A., Rosker Ch., James R.W., Rohr S., Kwak B.R..
The natural cardioprotective particle HDL modulates connexin43 gap junction channels. Cardiovasc. Res. 93:41-49, 2012 DOI
The natural cardioprotective particle HDL modulates connexin43 gap junction channels. Cardiovasc. Res. 93:41-49, 2012 DOI
M. Miragoli, Siti H., Kadir S. A., Sheppard M. N., Salvarani N., Virta M., Wells S., Lab M.J., Nikolaev V.O., Moshkov A., Hague W.M., Rohr S.,
Williamson C., Gorelik J..
A protective antiarrhythmic role of ursodeoxycholic acid in an InVitro rat model of the cholestatic fetal heart. HEPATOLOGY 54:1282-1292, 2011 DOI
A protective antiarrhythmic role of ursodeoxycholic acid in an InVitro rat model of the cholestatic fetal heart. HEPATOLOGY 54:1282-1292, 2011 DOI
C. Rosker, N. Salvarani, S. Schmutz, T. Grand, S. Rohr.
Abolishing myofibroblast arrhythmogeneicity by pharmacological ablation of a-smooth muscle actin containing stress fibers. Circ. Res. 109:1120-1131, 2011
Abolishing myofibroblast arrhythmogeneicity by pharmacological ablation of a-smooth muscle actin containing stress fibers. Circ. Res. 109:1120-1131, 2011
S. Rohr.
Cardiac fibroblasts in cell culture systems: Myofibroblasts all along?. J. Cardiovasc Pharmacol 57:389-399, 2011
Cardiac fibroblasts in cell culture systems: Myofibroblasts all along?. J. Cardiovasc Pharmacol 57:389-399, 2011
M Moridi, S. Tanner, N. Wyrsch, P.A. Farine, S. Rohr.
A highly sensitive a-Si photodetector array with integrated filter for optical detection in MEMS. Procedia Chemistry 1:1367-1370, 2009 DOI
A highly sensitive a-Si photodetector array with integrated filter for optical detection in MEMS. Procedia Chemistry 1:1367-1370, 2009 DOI
S. Rohr, M. Miragoli.
Cardiac electrophysiology: From cell to bedside. Cardiac myofibroblasts and arrhythmogenesis. In: Zipes DP, Jalife J, eds. 5th ed. Philadelphia:Saunders; 2009:269-278. 2009
Cardiac electrophysiology: From cell to bedside. Cardiac myofibroblasts and arrhythmogenesis. In: Zipes DP, Jalife J, eds. 5th ed. Philadelphia:Saunders; 2009:269-278. 2009
S. Rohr.
Cell to bedside: Myofibroblasts in diseased hearts: new players in cardiac arrhythmias?. Heart Rhythm 6:847-856, 2009 DOI
Cell to bedside: Myofibroblasts in diseased hearts: new players in cardiac arrhythmias?. Heart Rhythm 6:847-856, 2009 DOI
S. Rohr, M. Miragoli.
Cardiac myofibroblasts and arrhythmogenesis. In: Zipes DP, Jalife J, eds. Cardiac electrophysiology: From cell to bedside.5th ed. Philadelphia: Saunders; 2008:269-278. 2008
Cardiac myofibroblasts and arrhythmogenesis. In: Zipes DP, Jalife J, eds. Cardiac electrophysiology: From cell to bedside.5th ed. Philadelphia: Saunders; 2008:269-278. 2008
M. Helfenstein, M. Miragoli, S. Rohr, L. Mü, P. Wick, M. Mohr, P. Gehr, B. Rothen-Rutishauser.
Effects of combustion-derived ultrafine particles and manufactured nanoparticles on heart cells in vitro. Elsevier Toxicology 253:70-78, 2008 DOI
Effects of combustion-derived ultrafine particles and manufactured nanoparticles on heart cells in vitro. Elsevier Toxicology 253:70-78, 2008 DOI
V. Muñoz, K.R. Grzeda, T. Desplantez, S.V. Pandit, S. Mironov, S.M. Taffet, S. Rohr, A.G. Kléber, J. Jalife.
Adenoviral expression of IKs contributes to wavebreak and fibrillatory conduction in neonatal rat ventricular cardiomyocyte monolayers. Circ. Res. 101:475-483, 2007
Adenoviral expression of IKs contributes to wavebreak and fibrillatory conduction in neonatal rat ventricular cardiomyocyte monolayers. Circ. Res. 101:475-483, 2007
A.A Kondratyev, J.G.C. Ponard, A. Munteanu, S. Rohr, J.P. Kucera.
Dynamic changes of cardiac conduction during rapid pacing. Am. J. Physiol. 292: H1796-H1811, 2007
Dynamic changes of cardiac conduction during rapid pacing. Am. J. Physiol. 292: H1796-H1811, 2007
S. Rohr.
Molecular crosstalk between mechanical and electrical junctions at the intercalated disc. Circ. Res. 101:637-639 , 2007
Molecular crosstalk between mechanical and electrical junctions at the intercalated disc. Circ. Res. 101:637-639 , 2007
M. Miragoli, N. Salvarani, S. Rohr.
Myofibroblasts induce ectopic activity in cardiac tissue. Circ. Res. 101:755-758, 2007
Myofibroblasts induce ectopic activity in cardiac tissue. Circ. Res. 101:755-758, 2007
M. Miragoli, G. Gaudesius, S. Rohr.
Electrotonic modulation of cardiac impulse conduction by myofibroblasts. Circ. Res. 98:801-810, 2006
Electrotonic modulation of cardiac impulse conduction by myofibroblasts. Circ. Res. 98:801-810, 2006
L. Berdondini, P.D. van der Wal, O. Guenat, N.F. de Rooij, M. Koudelka-Hep, P. Seitz, R. Kaufmann, P. Metzler, N. Blanc, S. Rohr.
High-density electrode array for imaging in vitro electrophysiological activity. Biosens. Bioelectron. 21: 167-174, 2005
High-density electrode array for imaging in vitro electrophysiological activity. Biosens. Bioelectron. 21: 167-174, 2005
S. Rohr, J.P. Kucera.
Cardiac tissue architecture determines velocity and safety of propagation. DP. Zipes, J. Jalife eds. Cardiac Electrophysiology: From Cell To Bedside, 4th Edition 222 - 231, 2004
Cardiac tissue architecture determines velocity and safety of propagation. DP. Zipes, J. Jalife eds. Cardiac Electrophysiology: From Cell To Bedside, 4th Edition 222 - 231, 2004
S. Rohr.
Role of gap junctions in the propagation of the cardiac action potential. Cardiovasc. Res. 62: 309-322, 2004
Role of gap junctions in the propagation of the cardiac action potential. Cardiovasc. Res. 62: 309-322, 2004
G. Gaudesius, M. Miragoli, S.P. Thomas, S. Rohr.
Coupling of Cardiac Electrical Activity Over Extended Distances by Fibroblasts of Cardiac Origin. Circ. Res. 93:421-428, 2003
Coupling of Cardiac Electrical Activity Over Extended Distances by Fibroblasts of Cardiac Origin. Circ. Res. 93:421-428, 2003
S. Rohr, R. Flückiger-Labrada, J.P. Kucera.
Photolithographically defined deposition of attachment factors as a versatile method for patterning the growth of different cell types in culture. Pflugers Arch. 446:125-132, 2003
Photolithographically defined deposition of attachment factors as a versatile method for patterning the growth of different cell types in culture. Pflugers Arch. 446:125-132, 2003
J.P. Kucera, S. Rohr, Y. Rudy.
Localization of sodium channels in intercalated disks modulates cardiac conduction. Circ. Res. 91, No 12, 1176-1182, 2002
Localization of sodium channels in intercalated disks modulates cardiac conduction. Circ. Res. 91, No 12, 1176-1182, 2002
S. Rohr.
Optical mapping of microscopic impulse propagation. In:. Marcel Dekker Inc Quantitative Cardiac Electrophysiology (Eds. C. Cabo, D.S. Rosenbaum) pp. 507-554, 2002
Optical mapping of microscopic impulse propagation. In:. Marcel Dekker Inc Quantitative Cardiac Electrophysiology (Eds. C. Cabo, D.S. Rosenbaum) pp. 507-554, 2002
S. Rohr, J.P. Kucera.
Optical mapping of impulse propagation between cardiomyocytes. In: Opitcal Mapping of Cardiac Excitation and Arrhythmias (Eds. D.S. Rosenbaum, J. Jalife) Futura Pu pp. 113-135, 2001
Optical mapping of impulse propagation between cardiomyocytes. In: Opitcal Mapping of Cardiac Excitation and Arrhythmias (Eds. D.S. Rosenbaum, J. Jalife) Futura Pu pp. 113-135, 2001
A.G. Kléber, S. Rohr, V.G. Fast.
Role of cell-to-cell coupling, structural discontinuities, and tissue anisotropy in propagation of the electrical impulse. Futura Publishing Co. Inc. Armonk, NY In: Opitcal Mapping of Cardiac Excitation and Arrhythmias (Eds. D.S. Rosenbaum, J. Jalife) Futura Pu 137-155, 2001
Role of cell-to-cell coupling, structural discontinuities, and tissue anisotropy in propagation of the electrical impulse. Futura Publishing Co. Inc. Armonk, NY In: Opitcal Mapping of Cardiac Excitation and Arrhythmias (Eds. D.S. Rosenbaum, J. Jalife) Futura Pu 137-155, 2001
J.P. Kucera, A.G. Kléber, S. Rohr.
Slow conduction in cardiac tissue: Insights from optical mapping at the cellular level. J. Electrocardiol. 34 suppl 1:57-64, 2001
Slow conduction in cardiac tissue: Insights from optical mapping at the cellular level. J. Electrocardiol. 34 suppl 1:57-64, 2001
A.M. Gillis, V.G. Fast, S. Rohr, A.G. Kléber.
Mechanism of ventricular defibrillation. The role of tissue geometry in the changes in transmembrane potential in patterned myocyte cultures. Circulation 101:2438-2445, 2000
Mechanism of ventricular defibrillation. The role of tissue geometry in the changes in transmembrane potential in patterned myocyte cultures. Circulation 101:2438-2445, 2000
V.G. Fast, S. Rohr, R.E. Ideker.
Nonlinear changes of transmembrane potential caused by defibrillation shocks in strands of cultured myocytes. Am. J. Physiol. 278:H688-H697, 2000
Nonlinear changes of transmembrane potential caused by defibrillation shocks in strands of cultured myocytes. Am. J. Physiol. 278:H688-H697, 2000
J.P. Kucera, M.O. Heuschkel, P. Renaud, S. Rohr.
Power-law behavior of beat-rate variability in monolayer cultures of neonatal rat ventricular myocytes. Circ. Res. 86:1140-1145, 2000
Power-law behavior of beat-rate variability in monolayer cultures of neonatal rat ventricular myocytes. Circ. Res. 86:1140-1145, 2000
A.G. Kléber, J.P. Kucera, S. Rohr.
Principles of slow and discontinuous conduction. Experimental observations. In: Magalev T, Tchou JT, eds. Atrial av nodal electrophysiology: A view from the Millenium. Armonk, NY: Futura Publishing Co; 2000:73-88. 2000
Principles of slow and discontinuous conduction. Experimental observations. In: Magalev T, Tchou JT, eds. Atrial av nodal electrophysiology: A view from the Millenium. Armonk, NY: Futura Publishing Co; 2000:73-88. 2000
A.G. Kléber, V.G. Fast, S. Rohr.
Continuous and discontinuous propagation. In: Zipes DG, Jalife J, eds. Cardiac electrophysiology: From cell to bedside. Philadelphia: W.B.Saunders; 1999:205-213. 1999
Continuous and discontinuous propagation. In: Zipes DG, Jalife J, eds. Cardiac electrophysiology: From cell to bedside. Philadelphia: W.B.Saunders; 1999:205-213. 1999
S. Rohr, A.G. Kléber, J.P. Kucera.
Optical recording of impulse propagation in designer cultures. Cardiac tissue architectures inducing ultra-slow conduction. Trends in Cardiovasc Med 9:173-179, 1999
Optical recording of impulse propagation in designer cultures. Cardiac tissue architectures inducing ultra-slow conduction. Trends in Cardiovasc Med 9:173-179, 1999
V.G. Fast, S. Rohr, A.M. Gillis, A.G. Kléber.
Activation of cardiac tissue by extracellular electrical shocks: Formation of ’secondary sources’ at intercellular clefts in monolayers of cultured myocytes. Circ. Res. 82:375-385, 1998
Activation of cardiac tissue by extracellular electrical shocks: Formation of ’secondary sources’ at intercellular clefts in monolayers of cultured myocytes. Circ. Res. 82:375-385, 1998
S. Rohr, J.P. Kucera.
Optical recording system based on a fiber optic image conduit: Assessement of microscopic activation patterns in cardiac tissue. Biophys. J. 75:1062-1075, 1998
Optical recording system based on a fiber optic image conduit: Assessement of microscopic activation patterns in cardiac tissue. Biophys. J. 75:1062-1075, 1998
S. Rohr, J.P. Kucera, A.G. Kléber.
Slow conduction in cardiac tissue, i: Effects of a reduction of excitability versus. A reduction of electrical coupling on microconduction. Circ. Res. 83:781-794, 1998
Slow conduction in cardiac tissue, i: Effects of a reduction of excitability versus. A reduction of electrical coupling on microconduction. Circ. Res. 83:781-794, 1998
J.P. Kucera, A.G. Kléber, S. Rohr.
Slow conduction in cardiac tissue, ii: Effects of branching tissue geometry. Circ. Res. 83:795-805, 1998
Slow conduction in cardiac tissue, ii: Effects of branching tissue geometry. Circ. Res. 83:795-805, 1998
S. Rohr, J.P. Kucera, A.G. Kléber.
Form and function: Impulse propagation in designer cultures of cardiomyocytes. News Physiol Sci 12:171-177, 1997
Form and function: Impulse propagation in designer cultures of cardiomyocytes. News Physiol Sci 12:171-177, 1997
S. Rohr, J.P. Kucera.
Involvement of the calcium inward current in cardiac impulse propagation: Induction of unidirectional conduction block by nifedipine and reversal by bay k 8644. Biophys. J. 72:754-766, 1997
Involvement of the calcium inward current in cardiac impulse propagation: Induction of unidirectional conduction block by nifedipine and reversal by bay k 8644. Biophys. J. 72:754-766, 1997
A.G. Kléber, V.G. Fast, S. Rohr.
Microscopic conduction in cell cultures assessed by high-resolution optical mapping and computer simulation. Armonk, NY: Futura Publishing Company, Inc. 1997
Microscopic conduction in cell cultures assessed by high-resolution optical mapping and computer simulation. Armonk, NY: Futura Publishing Company, Inc. 1997
S. Rohr, J.P. Kucera, V.G. Fast, A.G. Kléber.
Paradoxical improvement of impulse conduction in cardiac tissue by partial cellular uncoupling. Science 275:841-844, 1997
Paradoxical improvement of impulse conduction in cardiac tissue by partial cellular uncoupling. Science 275:841-844, 1997
A.G. Kléber, V.G. Fast, J.P. Kucera, S. Rohr.
Physiologie und Pathophysiologie der kardialen Erregungsleitung. Zeitschrift für Kardiologie 85:25-33, 1996
Physiologie und Pathophysiologie der kardialen Erregungsleitung. Zeitschrift für Kardiologie 85:25-33, 1996
A.M. Gillis, V.G. Fast, S. Rohr, A.G. Kléber.
Spatial changes in transmembrane potential during extracellular electrical shocks in cultured monolayers of neonatal rat ventricular myocytes. Circ. Res. 79:676-690, 1996
Spatial changes in transmembrane potential during extracellular electrical shocks in cultured monolayers of neonatal rat ventricular myocytes. Circ. Res. 79:676-690, 1996
S. Rohr.
Determination of impulse conduction characteristics at a microscopic scale in patterned growth heart cell cultures using multiple site optical recording of transmembrane voltage. J. Cardiovasc. Electrophysiol. 6:551-568, 1995
Determination of impulse conduction characteristics at a microscopic scale in patterned growth heart cell cultures using multiple site optical recording of transmembrane voltage. J. Cardiovasc. Electrophysiol. 6:551-568, 1995
S. Rohr, B.M. Salzberg.
Characterization of impulse propagation at the microscopic level across geometrically defined expansions of excitable tissue: Multiple site optical recording of transmembrane voltage (msortv) in patterned growth heart cell cultures. J. Gen. Physiol. 104:287-309, 1994
Characterization of impulse propagation at the microscopic level across geometrically defined expansions of excitable tissue: Multiple site optical recording of transmembrane voltage (msortv) in patterned growth heart cell cultures. J. Gen. Physiol. 104:287-309, 1994
S. Rohr, B.M. Salzberg.
Multiple site optical recording of transmembrane voltage in patterned growth heart cell cultures: Assessing electrical behavior, with microsecond resolution, on a cellular and subcellular scale. Biophys. J. 67:1301-1315, 1994
Multiple site optical recording of transmembrane voltage in patterned growth heart cell cultures: Assessing electrical behavior, with microsecond resolution, on a cellular and subcellular scale. Biophys. J. 67:1301-1315, 1994
S. Rohr, B.M. Salzberg.
Discontinuities in action potential propagation along chains of single ventricular myocytes in culture: Multiple site optical recording of transmembrane voltage (msortv) suggests propagation delays at the junctional sites between cells. Biol Bull Mar Biol Lab 183:342-343, 1992
Discontinuities in action potential propagation along chains of single ventricular myocytes in culture: Multiple site optical recording of transmembrane voltage (msortv) suggests propagation delays at the junctional sites between cells. Biol Bull Mar Biol Lab 183:342-343, 1992
A.L. Obaid, D.J. Zou, S. Rohr, B.M. Salzberg.
Optical recording with single cell resolution from a simple mammalian nervous system: Electrical activity in ganglia from the submucuous plexus of the guinea-pig ileum. Biol Bull Mar Biol Lab 183: 340-342, 1992
Optical recording with single cell resolution from a simple mammalian nervous system: Electrical activity in ganglia from the submucuous plexus of the guinea-pig ileum. Biol Bull Mar Biol Lab 183: 340-342, 1992
S. Rohr, D.M. Schölly, A.G. Kléber.
Patterned growth of neonatal rat heart cells in culture. Morphological and electrophysiological characterization. Circ. Res. 68:114-130, 1991
Patterned growth of neonatal rat heart cells in culture. Morphological and electrophysiological characterization. Circ. Res. 68:114-130, 1991
S. Rohr.
A computerized device for long term measurements of the contraction frequency of cultured rat heart cells under stable incubating conditions. Pflugers Arch. 416:201-206, 1990
A computerized device for long term measurements of the contraction frequency of cultured rat heart cells under stable incubating conditions. Pflugers Arch. 416:201-206, 1990
G. Schmilinsky, A. Rüdisüli, M Willi, S. Rohr, R. Weingart.
Effects of arachidonic acid on gap junctions of neonatal rat heart cells. Pflugers Arch. 417:149-156, 1990
Effects of arachidonic acid on gap junctions of neonatal rat heart cells. Pflugers Arch. 417:149-156, 1990
A. Koch, S. Rohr, J. Plaschkes et al..
Incidence of gastroesophageal reflux following repair of esophageal atresia. Prog. Ped. Surg. 19:103-113, 1986
Incidence of gastroesophageal reflux following repair of esophageal atresia. Prog. Ped. Surg. 19:103-113, 1986
